- API
- Ivabradine hydrochlorid
- Rivaroxaban
- Apixaban
- Dabigatran etexilate
- Safinamide
- Ticlopidine hydrochloride
- Mirtazapine
- Clopidogrel bisulfate
- Duloxetine hydrochloride
- Rebamipide
- Intermediates
- Duloxetine intermediate
- Clopidogrel intermediate
- Mirtazapine intermediates
- Dabigatran etexilate intermediates
- Lurasidone intermediates
- Bepotastine intermediates
- Asenapine Intermediates
- Drotaverine intermediates
- Prasugrel Intermediates
- Ivabradine、Verapamil, Denopamine intermediates
- Moxifloxacin intermediate
- Ticagrelor intermediate <Under Development>
- Canagliflozin intermediate
- Ivabradine intermediate
Address: Zhejiang Provincial Chemical and Medical Raw Material Base Linhai Zone, Duqiao Town, Linhai City, Zhejiang Province, 317016, China
Tel / Fax: 0576-85588211
Tel / Fax: 0576-85588211
| Product Name | Structure | CAS No. | Specification | Use | Classification |
| Ivabradine hydrochlorid | 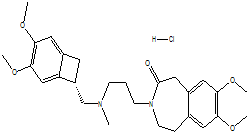 |
148849-67-6 | EU GMP | API | |
| Rivaroxaban | 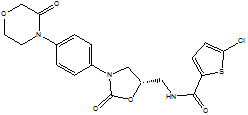 |
366789-02-8 | Lab study | API | |
| Apixaban | 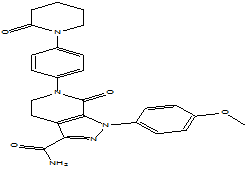 |
503612-47-3 | Lab study | API | |
| Dabigatran etexilate | 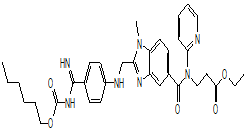 |
211915-06-9 | lab study | API | |
| Safinamide |  |
133865-89-1 | lab study | API | |
| Ticlopidine hydrochloride | 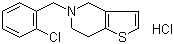 |
53885-35-1 | EP;JP | EU GMP/CEP/JP GMP | API |
| Mirtazapine | 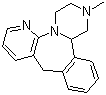 |
85650-52-8 | JP;EP | EU GMP/CEP/JP GMP | API |
| Clopidogrel bisulfate | 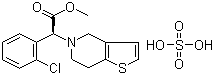 |
120202-66-6 | USP/EP | CEP/US DMF | API |
| Duloxetine hydrochloride | 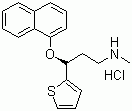 |
136434-34-9 | USP/EP | EU GMP/CEP | API |
| Rebamipide | 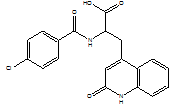 |
90098-04-7 | JP | JP GMP | API |



